Abstract
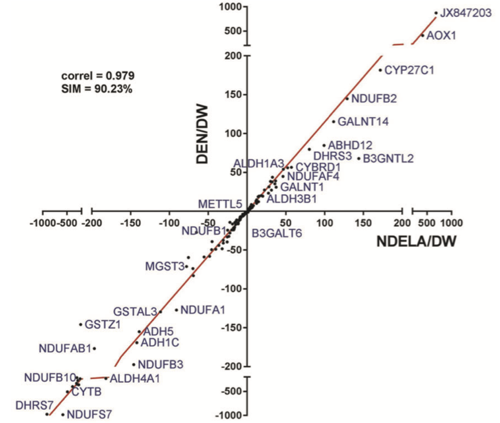
The Chicken Egg Genotoxicity Assay (CEGA) demonstrated responsiveness to various DNA-reactive chemicals requiring metabolic activation, which implies broad biactivation capability. To assess potential metabolic competence, expression profiles of metabolic genes in the embryo-fetal chicken livers were determined using microarray technology. Fertilized chicken eggs were injected under the CEGA protocol with vehicle (deionized water (DW)), the activation-dependent carcinogens, diethylnitrosamine (DEN), and N-nitrosodiethanolamine (NDELA) at doses producing no effect on survival. Previously in CEGA, DEN produced DNA damage, whereas NDELA did not. Expressions of 463 genes known to encode for phase I and II of endo- and xenobiotic metabolism were detected on the array. DW did not affect the expression of the selected genes, deregulating less than 1% of them. In contrast, DEN at 2 mg/egg and NDELA at 4 mg/egg produced significant transcriptomic alterations, up-regulating up to 41% and down-regulating over 31% of studied genes. Both nitrosamines modulated the majority of the genes in a similar manner, sharing 64 up-regulated and 93 down-regulated genes with respect to control group, indicating similarity in the regulation of their metabolism by avian liver. Differences in gene expression between DEN and NDELA were documented for several phase I CYP 450 genes that are responsible for nitrosamine biotransformation, as well as for phase II genes that regulate detoxication reactions. These findings could underlie the difference in genotoxicity of DEN and NDELA in CEGA. In conclusion, the analysis of gene expression profiles in embryo-fetal chicken livers dosed with dialkylnitrosamines demonstrated that avian species possess a complex array of inducible genes coding for biotransformation.